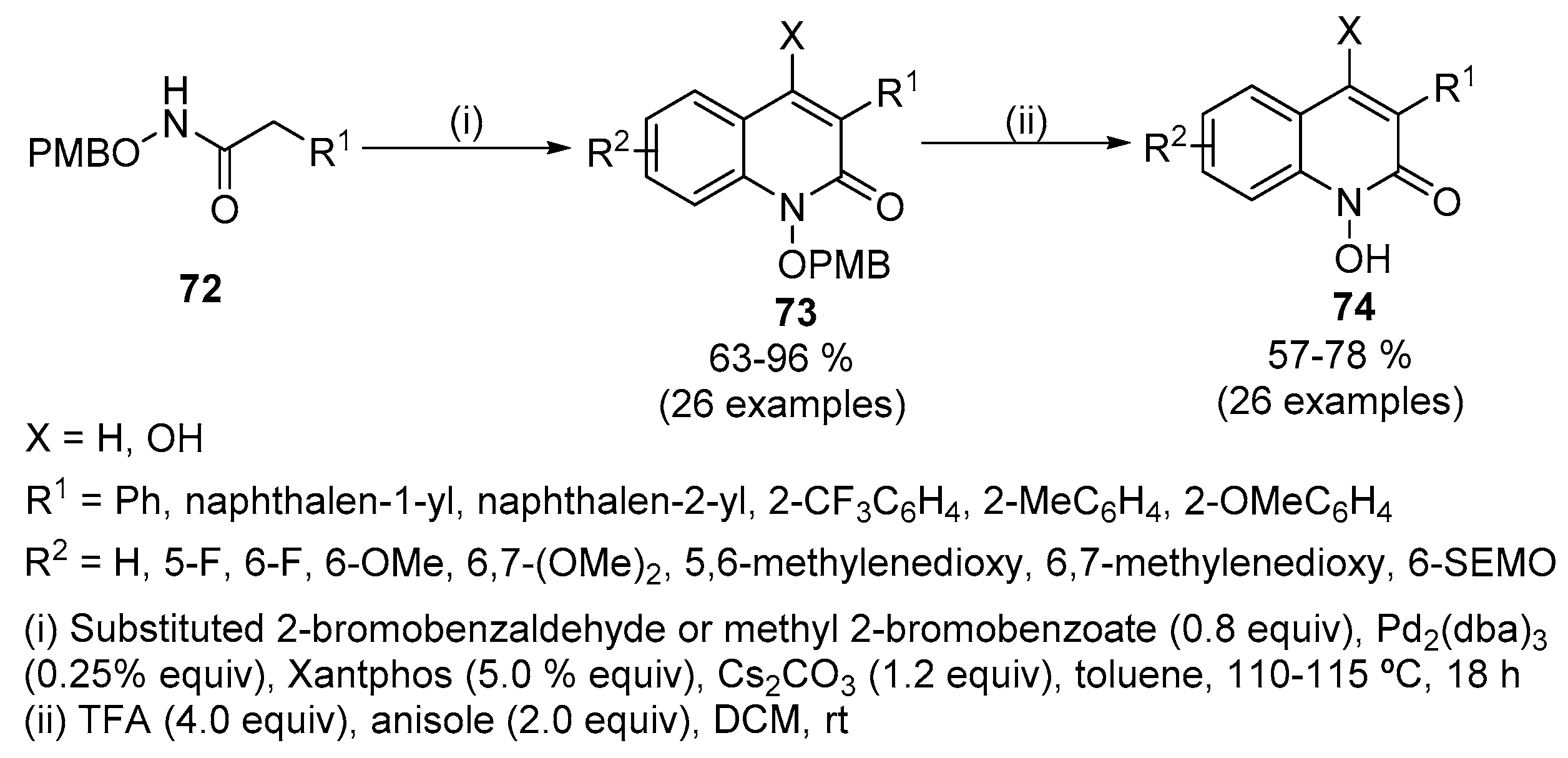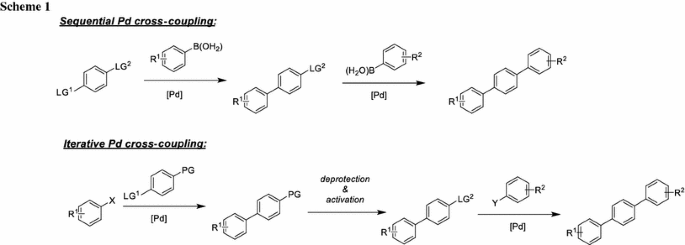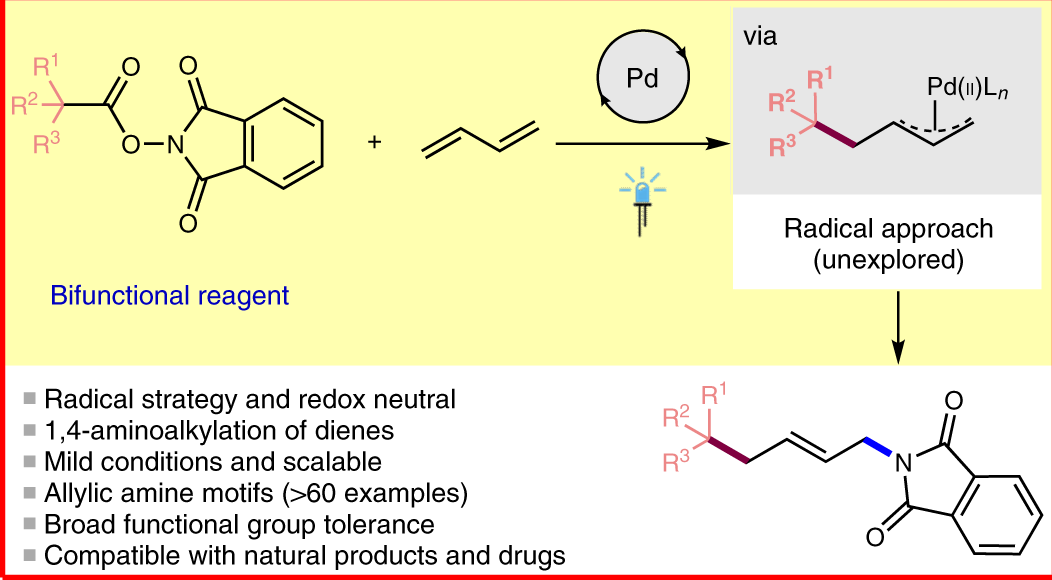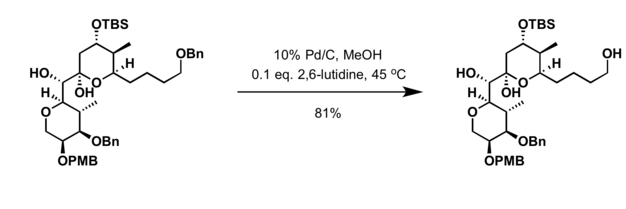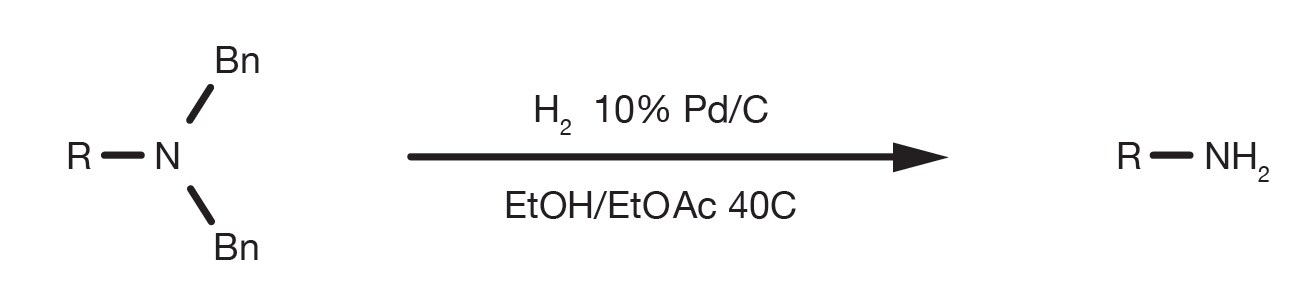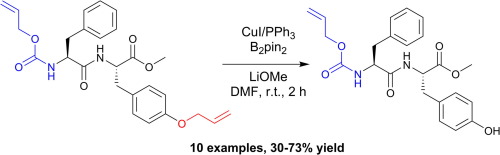
A mild copper catalyzed method for the selective deprotection of aryl allyl ethers,Tetrahedron Letters - X-MOL
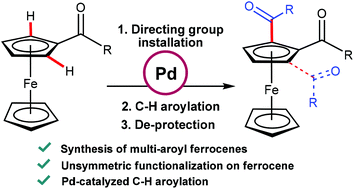
Synthesis of unsymmetrical multi-aroyl derivatives of ferrocene using palladium catalysed oxidative C–H aroylation - Dalton Transactions (RSC Publishing)

Catalytic Hydrogenation: Catalytic Reduction of Pd/C, Deprotection and Lindlar Catalyst | Ryosuke University

Intracellular Deprotection Reactions Mediated by Palladium Complexes Equipped with Designed Phosphine Ligands. - Abstract - Europe PMC
Intracellular Deprotection Reactions Mediated by Palladium Complexes Equipped with Designed Phosphine Ligands

Molecules | Free Full-Text | Nickel-Catalyzed Removal of Alkene Protecting Group of Phenols, Alcohols via Chain Walking Process | HTML
A Mechanistic Study of Direct Activation of Allylic Alcohols in Palladium Catalyzed Amination Reactions

Low-valent titanium-catalyzed deprotection of allyl- and propargyl-carbamates to amines - ScienceDirect
Intracellular Deprotection Reactions Mediated by Palladium Complexes Equipped with Designed Phosphine Ligands

Optimization of reaction conditions for deprotection along with cross... | Download Scientific Diagram

Palladium-catalyzed reaction of tributyltin hydride. Selective and very mild deprotection of allyl and allyloxycarbonyl derivatives of amino-acids

Pd(II)-catalyzed deprotection of acetals and ketals containing acid sensitive functional groups - ScienceDirect
Intracellular Deprotection Reactions Mediated by Palladium Complexes Equipped with Designed Phosphine Ligands