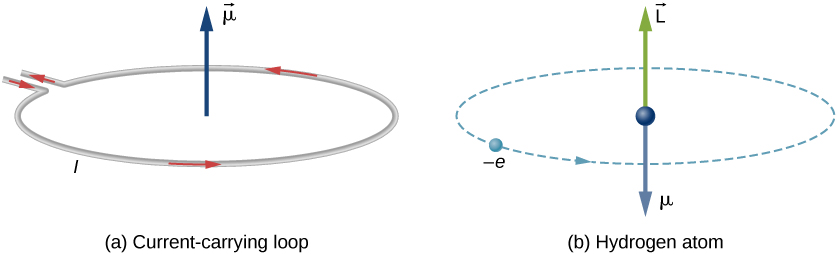
In the Bohr model an electron moves in a circular orbit around the proton. Considering the orbiting electron to be circular current loop, the magnetic moment of the hydrogen atom, when 5th
Define the term magnetic moment of current loop. Derive the expression forthe magnetic moment when an electron revolves at a speed 'v' around an orbitof radius r in hydrogen atom. Also calculate
Prove that the magnetic moment of the electron revolving around anucleus in an orbit of radius r with orbital speed v is equal to evr/2.Hence using Bohr's postulate of quantization of angular

42. The ratio of magnetic dipole moment of an electron of charge e and mass m in Bohr's orbit in hydrogen atom to its angular momentum is :
Magnetic Moment Physics Of Magnetic Resonance Imaging Magnetic Field Hydrogen Atom, PNG, 1233x874px, Magnetic Moment, Area,

Three protons coming from excited atomic hydrogen sample are picked up. Their energies are 12.1 eV, 10.2 eV and 1.9 eV . These photons must come from

In the Bohr model of the hydrogen atom, the electron circuulates around the nucleus in a path of radius 5xx10^
In a hydrogen atom, an electron of charge revolves in an orbit of radius r with speed v. What is the magnitude of the resulting magnetic moment of the electron? - Quora